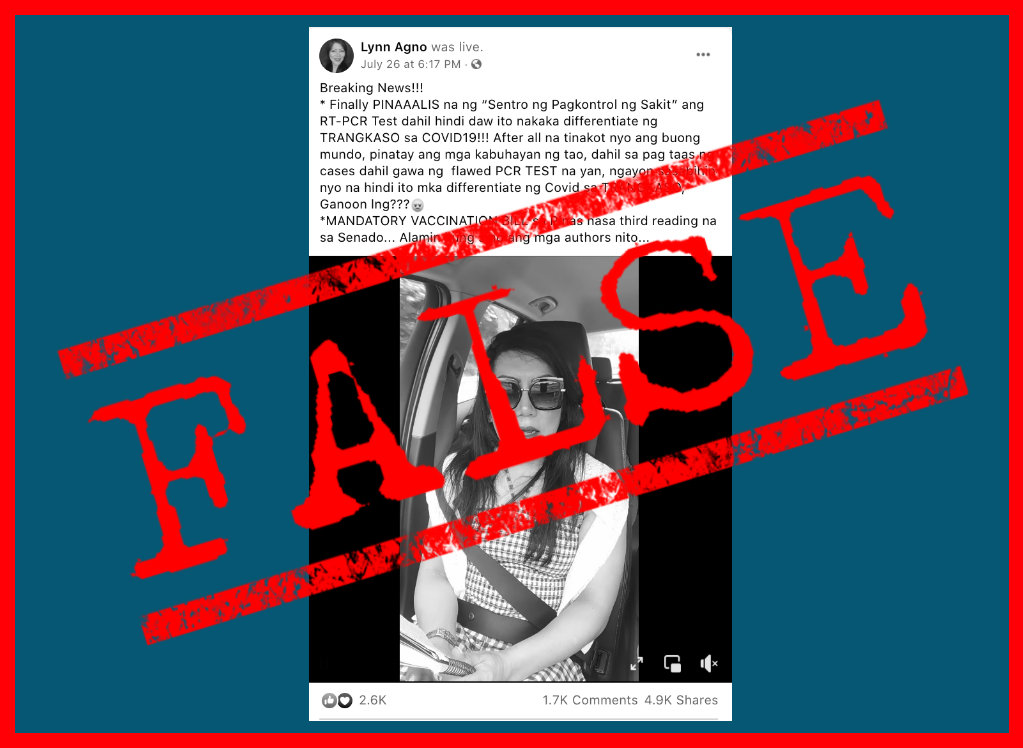
An April 8 report about the temporary suspension of the Oxford-AstraZeneca COVID-19 vaccine in the Philippines is being peddled on social media as current news, even though the suspension was already lifted in early May.
The misleading video bore text questioning why the Department of Health (DOH) is only taking action now “when a lot of people have already been vaccinated” with AstraZeneca. The posts failed to mention that the news clip is nearly two months old, and that the DOH has already resumed the inoculation of Vaxzevria for all age groups since May 7.
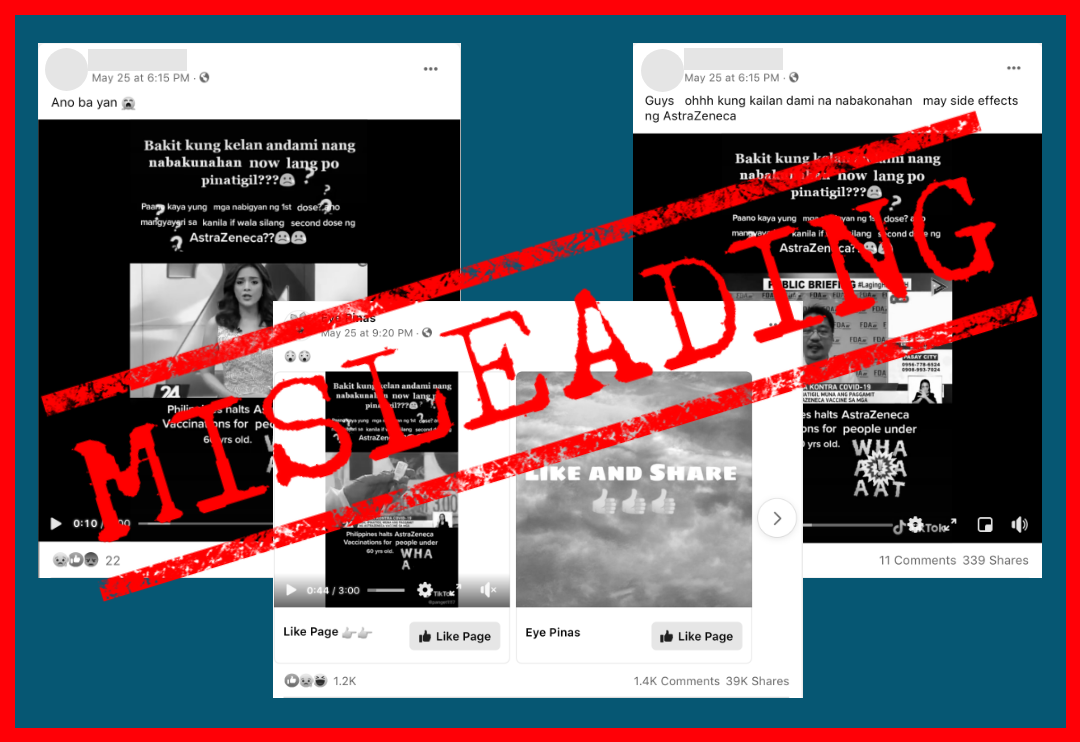
This now deleted three-minute TikTok video by user panget1117 was reposted by at least two netizens and one Facebook (FB) page — Eye Pinas — on May 25 . It was a video clip from a five-minute news report of the news program 24 Oras that aired on April 8, about the DOH’s decision to temporarily halt the use of AstraZeneca jabs on individuals below 60 years old.
DOH’s initial suspension of the vaccine was in accordance with the recommendation of the Food and Drug Administration (FDA) following reports of rare cases of blood clotting (thrombosis) and low platelet count (thrombocytopenia) among individuals who received AstraZeneca jabs in Europe.
The European Medicines Agency (EMA) said on April 7 that the mentioned adverse reactions, which usually occur within two weeks after vaccination, “should be listed as very rare side effects of Vaxzevria.”
However, EMA pointed out in an April 23 follow-up assessment report on the vaccine that thrombosis and thrombocytopenia “[have] been characterised as very rare” events in terms of frequency in reported cases. The World Health Organization puts the ratio at four cases per one million vaccinated adults, based on the data it acquired from the United Kingdom.
EMA also emphasized that the overall benefits of the AstraZeneca vaccine remain to “outweigh the risks of side effects” of COVID-19.
This was also echoed by the DOH in its May 7 announcement lifting the temporary suspension on Vaxzevria, with the department’s All Experts Group and the Philippine College of Hematology and Transfusion Medicine (PCHTM) saying that “there are currently no known risk factors for Vaccine-Induced Thrombosis and Thrombocytopenia (VITT).”
The DOH stressed that there hasn’t been a single VITT case recorded in the Philippines to date. The PCHTM also released updated guidelines on managing adverse events following immunization, including ensuring that all vaccination sites in the country will have “a strengthened post-vaccination surveillance”, and that healthcare workers will be trained to recognize and manage symptoms of VITT.
The misleading posts started circulating on FB just a week after the DOH — as ordered by President Rodrigo Duterte — encouraged local government units to not announce what brands they will administer in their COVID-19 vaccination programs, to avoid the flocking of people in inoculation sites where their preferred brand will be given.
The three copies of the misleading video have received over 1.7 million collective views, 1,400 reactions, 1,800 comments, and 48,500 shares from netizens.
(Editor’s Note: VERA Files has partnered with Facebook to fight the spread of disinformation. Find out more about this partnership and our methodology.)