A man who produced a video in which he claims COVID-19 vaccines are unsafe, is using a statement made by President Rodrigo Duterte over four years ago as “proof” that vaccines are part of a scheme to reduce the Filipino population.
This is false. Duterte’s speech was used in the wrong context.
In an Aug. 4 Facebook (FB) post, the Presidential Communications Operations Office called the 17-minute video “fake news.”
The video, which has been grabbed, reuploaded and shared on Facebook (FB) by several people since July 27, featured Rey Valeros, who describes himself as a Navy reserve officer. He started hosting a weekly segment called On the Ground on the Facebook page of the political party Katipunan ng Demokratikong Pilipino in July.
Valeros, who has no clear background in medicine, has been pushing for the use of a drug that he claims can cure COVID-19 and “all major viral diseases.”
In the circulating video, Valeros featured an incorrect claim by the president’s chief legal counsel Salvador Panelo that vaccinated Filipinos are like “guinea pigs.” He also wrongly said that Johnson & Johnson’s COVID-19 vaccine has not undergone proper testing.
VERA Files Fact Check dissected these claims and debunked them below:
On Duterte’s statement
Screenshot of Valeros’ video.
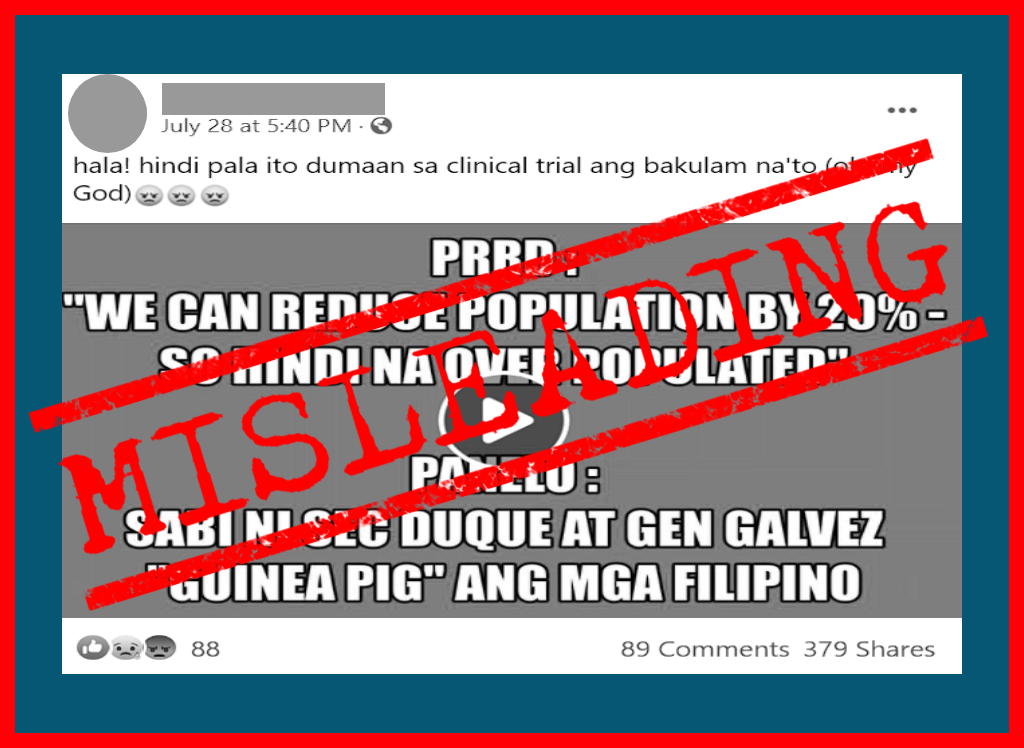
To support his claim that the government is sacrificing the lives of Filipinos for vaccines against the coronavirus disease 2019 (COVID-19), Valeros used a 12-second clip from a 57-minute speech delivered by Duterte at the Davao Investment Conference on July 23, 2017.
Duterte was shown saying: “There is too much of us, 110 million. We can sacrifice about 20. So, it becomes 80. So hindi na (we don’t become) overpopulated.”
The president was talking about eliminating members of the Communist Party of the Philippines whom he called “enemies of the state.”
On Panelo’s ‘guinea pig’ statement

Screenshot of Valeros’ video.
Valeros also showed a clip from a March 23 episode of Panelo’s talk show Counterpoint, where the former Palace spokesperson commented on the statements by Health Secretary Francisco Duque III and vaccine czar Carlito Galvez Jr. that the slow rollout of COVID-19 vaccines at the time was due to concerns over their side effects.
Panelo added that “not all of the doses were being rolled out” because the first vaccine recipients were “basically, guinea pigs.” This is misleading.
In March, Duque and Galvez did say vaccination rollout in the country had been slow because of the observance of adverse events. But neither official said Filipinos were being used as guinea pigs for vaccine experimentation.
In a March 12 interview on the GMA News program 24 Oras, 12 days into the country’s rollout of COVID-19 vaccines, Duque attributed the slowdown to bottlenecks in observation areas for adverse events, where people who have received a jab are monitored between 30 minutes and an hour.
Four days before, Galvez told Duterte in a Cabinet meeting that Duque ordered a “calibrated and measured” rollout of the Astrazeneca vaccines due to reports of risks for adverse events.
On J&J; vaccines
|
Claim |
Rating |
|
The vaccines didn’t undergo animal trials, they were immediately tried on humans. |
False |
J&J;’s one-and-done vaccine, Ad26.COV2.S, had been tested on several animal species including monkeys, hamsters, rabbits, and mice.
One of the first studies that showed the drug could protect against SARS-CoV-2, the virus causing COVID-19, involved 52 rhesus macaques, a monkey species. Published in July 2020 in the scientific journal Nature, it observed that the vaccine provided complete and near-complete protection against the novel coronavirus.
Ad26.COV2.S was also tested in golden Syrian hamsters. A study published in September 2020in the Nature Medicine journal showed it could protect against severe COVID-19 infection in the hamsters, and a March 2021 article published in Nature Partner Journals showed it could protect the animals against the G614 variant of SARS-CoV-2.
On COVID-19 vaccines and clinical trials
|
Claim |
Rating |
|
COVID-19 vaccines are being rolled out, but they’re still undergoing testing. |
Needs Context |
To speed up immunization of people against a public health crisis such as COVID-19, vaccines currently undergoing clinical trials can be granted a temporary emergency use authorization (EUA) provided their benefits outweigh known and potential risks, and that no adequate, approved and available alternatives currently exist.
The Philippine FDA grants an EUA to a COVID-19 vaccine based on data from adequate and well-controlled clinical trials. FDA Director General Eric Domingo has said that the Philippines will only accept EUA applications for vaccines or drugs that have been granted an EUA by similar regulatory authorities abroad such as those in the U.S. and European Union, among others.
The U.S. FDA allows vaccine manufacturers to submit an EUA request based on partial or complete results of a phase 3 clinical trial, and safety data from phase 1 and 2 studies. EUA requests are expected to also include a safety database of over 3,000 phase 3 participants who have been monitored for adverse effects.
The first phase of clinical trials tests a new drug on tens of people to determine a safe dosage range and identify side effects. A phase 2 trial evaluates a drug’s effectiveness to hundreds of people and again its safety, while phase 3 is conducted on thousands of people to confirm the drug’s effectiveness and gather more data on long-term and rare side effects, according to the U.S. FDA.
On COVID-19 vaccine related deaths, adverse effects
|
Claim |
Rating |
|
Hundreds of people have gotten sick from the vaccines |
Needs Context |
|
Some died from vaccine side effects |
No Basis |
From March 1 to Aug. 1, a total of 53,629 suspected adverse reaction reports (0.26%) out of 20,863,544 administered doses were received, evaluated, and analyzed by the Philippine FDA, according to its latest report.
Of the reported adverse reactions, 52,190 (97.32%) were non-serious events such as high blood pressure, headache and fever, while 1,436 (2.68%) were serious events such as severe allergic reaction, hospitalization or increased hospital stay, and rare blood clotting. The FDA says these serious events are being reviewed to see if the vaccine was the cause.
There were also 526 reported cases of death but the FDA says this does not necessarily mean they were caused by the vaccine. Of the total deaths, 231 have been ruled as coincidental or not related to the vaccine, 17 cases were “indeterminate,” while 12 cases were “unclassifiable.” The rest are still under investigation.
Three netizens have posted the misleading video, garnering over 160 reactions, 130 comments, and 5,700 views on FB. An earlier, more viral copy posted by a Facebook user on July 20 and taken down as of Aug. 2, emerged four days after the country received over 3.2 million doses of J&J; vaccines donated by the United States.
(Editor’s Note: VERA Files has partnered with Facebook to fight the spread of disinformation. Find out more about this partnership and our methodology.)




