Despite the loud applause on his call for a “stronger healthcare system” during his first State of the Nation Address (SONA) on Monday, President Ferdinand Marcos Jr. remained silent on his plans for the regulation of novel tobacco products.
Marcos did not heed the call of former health secretaries, heads of health agencies, and several medical groups to veto the contested vape bill and instead allowed it to lapse into law on July 25. (See Health expert counts on Marcos Jr. to protect youth from tobacco, vape harm)
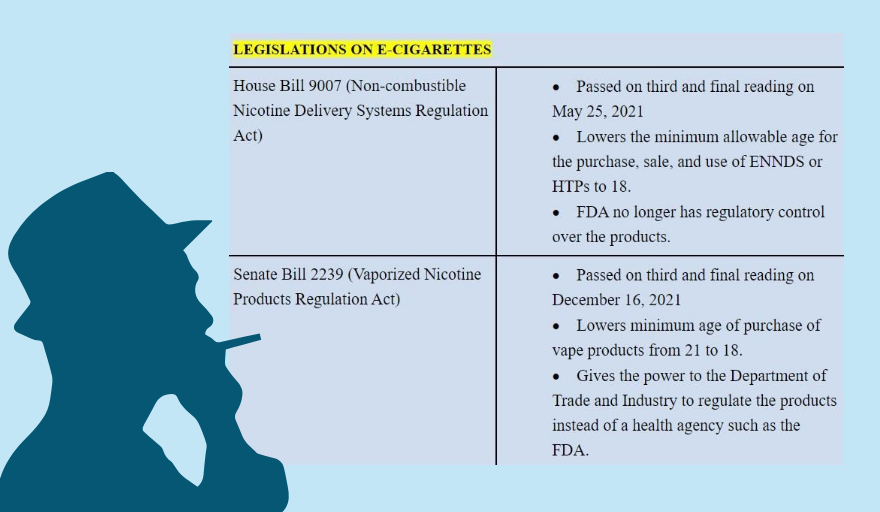
Congress ratified the proposed Vaporized Nicotine and Non-Nicotine Products Regulation Act on Jan. 26, but the House of Representatives transmitted it to Malacañang only on June 24, just six days before then-president Rodrigo Duterte, who is anti-tobacco, stepped out of office. (See Vape bill version 2022: Congress ‘hijacks’ stringent regulations)
Public interest law group ImagineLaw described the measure as a “betrayal of public health.” In a statement on July 26, ImagineLaw Executive Director Sophia San Luis said the passage of the law is a “regrettable development in the face of President Ferdinand ‘Bongbong’ Marcos Jr.’s commitment to build back better.”
The new measure lowers the age of persons who can buy, use and sell electronic nicotine and non-nicotine devices (ENNDS) and heated tobacco products (HTPs) from 21 to 18 years old and allows a range of flavors in the market other than plain tobacco and plain menthol. (See Bill provides younger people easier access to smoke-free alternatives)
“It dismantles existing measures that protect our health and protect our youth from lifelong addiction,” San Luis said, adding that it is a “deregulation measure that will lead to a vaping epidemic among young people.”
Dr. Rizalina Gonzalez, chair of the Tobacco Control Committee in the Philippine Pediatric Society, has stressed that smoking is a pediatric disease and many smokers start smoking before 18 years old. (See VERA FILES FACT SHEET: What lowering age restrictions on e-cigarette smoking means for the youth)
“[Vaping] really targets the children… When I didn’t know anything about vaping, I thought the flavoring was for cake. But now that I look at it, being next to the lighter, it really is deceiving,” Gonzalez said in a previous interview with VERA Files.
(See Vape bill eases restrictions on flavors, online advertising and Vape bill exposes young ‘vulnerable’ consumers to health risks, endangers already weak enforcement)
The law eases the strict provisions regulating vapes and HTPs in Republic Act (RA) 11467, a tax measure on alcohol and e-cigarettes that includes regulatory provisions on novel tobacco products. RA 11467 took full effect just this May, after an 18-month transition period from its enactment.
It transfers the sole authority to regulate vapes and HTPs from the Food and Drug Administration (FDA) to the Department of Trade and Industry (DTI) by reclassifying it as a consumer item, instead of a health product.
In a July 26 press release, Sen. Pia Cayetano said she had high hopes Marcos would take the side of health advocates, especially when he “repeatedly emphasized that science will be a cornerstone of his administration’s programs, including in health and education.”
“I thought this means that the vape bill would be vetoed,” Cayetano said, “because the science clearly tells us just how harmful these products are.”
“Medical experts have repeatedly said how the vape bill masquerades as a health measure, as it really pushes for deregulation, not regulation, and harm introduction, not harm reduction,” she added.
Meanwhile, some pro-tobacco groups and medical practitioners have been promoting vapes and HTPs as “less harmful” than conventional tobacco smoking and could help smokers “quit.”
Cayetano was one of two legislators who dissented from the passage of the bill in the Senate on Dec. 16, 2021. (See How Congress voted on the vape bill)
“If we can allow a product that clearly has effects on health, whether harmful, not so harmful, very harmful… What’s next? Where will the next lobby come from?” Cayetano said in a privilege speech during the first regular session of the 19th Congress.
Recently, the Supreme Court (SC) affirmed that the FDA must be in charge of the “health aspects” of tobacco products, even if Section 25 of RA 9211 or the Tobacco Regulation Act of 2003 gave the Inter-Agency Committee Tobacco (IAC-T) the “exclusive jurisdiction over tobacco products, including its health aspect.”
Justice Marvic Leonen, who penned the SC ruling, said the IAC-T has “limited jurisdiction over tobacco products and does not regulate all their aspects.” He added that RA 9211 covers only regulation of distribution, access, sale, labeling, advertisements, sponsorships, and promotions of tobacco products.
The Philippine Tobacco Institute (PTI), which represents major transnational tobacco companies, was suggesting an interpretation of the law that removes them from the control of the FDA and the Department of Health (DOH), Leonen said.
(See DOH, FDA win in SC could go to waste if vape bill becomes law and SC ruling has ‘far-reaching’ effects on tobacco regulation –health advocates)
Read VERA Files’ full coverage here: How the tobacco industry interferes in policy making